Non-hereditary Myopathies
Idiopathic inflammatory myopathies
6. Paraneoplastic ( cancer-associated ) myositis
Several previous reports have described higher rates of association of cancer with DM than those observed in the normal population (Fig. 47).
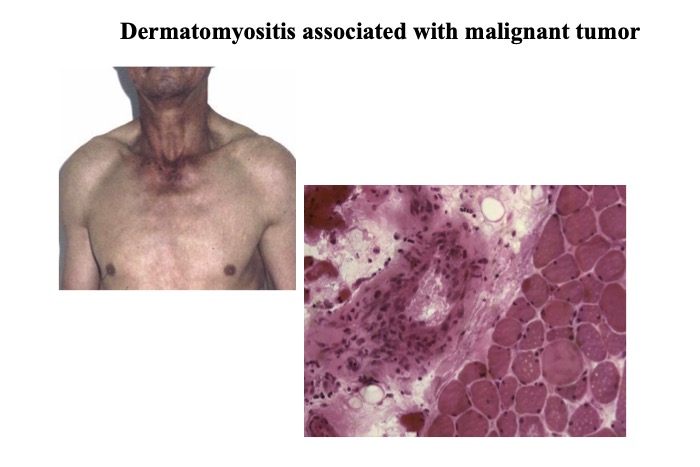
Fig.47
Skin rash in the V neck region of a patient with paraneoplastic myositis. A limited degree of inflammation is seen around the blood vessels in the perimysium.
The frequency of association varied based on the patient groups studied and ranged between 9 and 42%. Clinical features of DM showing a high rate of association with cancer (risk factors) included advanced age, male sex, severe skin changes with necrosis or ulcers resistant to treatment, histopathological changes of vasculitis with leukoclastic phenomenon observed in muscle specimens, and involvement of distal, respiratory or bulbar muscles. In contrast, features of DM showing a low rate of association with cancer (non-risk factors) included interstitial pneumonitis, arthropathy, Raynaud phenomenon, cardiomyopathy, fever, anti-nuclear antigen positivity on serological examination, and lymphopenia, all suggesting an association with connective tissue disease (Zahr, 2011).
In contrast to DM, PM has been reported to be unassociated with cancer. However, a moderate but significant association (5–18%) was reported by some studies (Sigurgeirsson, 1992). A study from Australia reported that the rate of association with cancer was 2-fold (1.4–2.7)higher than that in the general population in patients diagnosed with PM based on muscle biopsy (Buchbinder, 2001). Studies from Taiwan have reported that patients with PM show a higher rate of association with cancer, particularly nasopharyngeal carcinoma (Huang, 2009). Data regarding JDM are sparse in the available literature; however, a low association with malignancy is reported. A limitation of these studies was that in many of these studies the diagnosis of PM was based on the Bohan and Peter (1975ab) criteria, which might not have excluded probable DM without skin rash (Dalakas, 2003) and immune mediated necrotizing myopathies (IMNM).
Detection of antibodies is important for the diagnosis of cancer-associated myositis (CAM). A high association with cancer is reported in patients with IMNM, both in patients with anti-signal recognition particle (SRP) and anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR) antibodies. Targoff et al. (2006) reported an antibody against a protein of molecular weight 155 kDa, which they proposed was a transcriptional intermediary factor-1 gamma (TIF1γ). To date, this antibody remains the most important marker for CAM with sensitivity of 78% and specificity of 89% (Trallero-Araguas, 2012). The anti-nuclear matrix protein 2 (NXP2) antibody shows a positivity rate of 17% compared with 38% of the anti-TIF1-γ antibody (Fiorentino, 2013). In Japan, the anti-NXP2 antibody showed a positivity rate of 1.6% in patients with DM and PM. An association with cancer was observed in 29% of patients with DM with a positive anti-NXP2 antibody (Ichimura, 2012).
Usually, CAM is indistinguishable from DM without cancer solely based on histopathological examination of muscle specimens. However, CAM tends to show insignificant inflammation and frequent necrotic fibers in both perifascicular and endomysial regions. Several case reports have described IBM associated with cancer, although whether the association rate is significantly higher than that observed in the general population remains unconfirmed.




