Muscle biopsy
Basic histopathological changes in muscle
Assessment of histopathological changes in muscle involves attention to the following indicators: changes in muscle fibers can be easily identified by observing the diameter of muscle fibers. Muscle fibers may be atrophic, normal, or hypertrophic. Shape of the atrophic fibers and their distribution can provide important clues regarding the etiology of the atrophic changes. Frequency of internal nuclei and other morphological features of individual muscle fibers are also informative. Histochemical analysis provides additional information regarding the internal structure of muscle fibers and any abnormalities (increase or decrease) of the individual components of muscle fibers. Analysis of changes in fiber types is useful to determine issues associated with innervation and re-innervation of muscle fibers, as well as issues associated with development and regeneration. Observing the interstitial tissue provides valuable details regarding inflammation, changes in the blood vessels and intramuscular nerves fibers, fibrosis, and fat deposition. Muscle spindles and neuromuscular junctions are identified at specific locations. Furthermore, immunohistochemical studies are playing an increasingly important role as a useful diagnostic aid in recent years.
1.Neurogenic muscle atrophy
Impairment of the anterior horn cells (motor neurons), anterior roots of the spinal cord or peripheral nerves leads to neurogenic atrophy of muscle fibers. Histopathological changes in muscle are often represented by “grouped atrophy” and “angulated fibers”. The former refers to atrophy of groups of muscle fibers in which muscle fibers belonging to the same motor unit are simultaneously affected. The number of fibers affected within a group varies greatly (from less than ten to hundreds) (Fig.7).
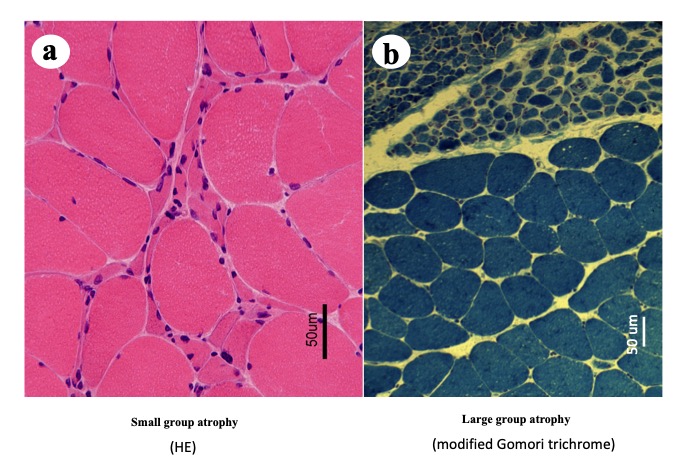
Fig.7
Small (a) and large (b) group atrophy
During early phase of neurogenic atrophy, the muscle specimen shows a pattern of scattered small groups of atrophic fibers, whereas during the later stages, large groups of atrophic fibers are predominant. With regard to fiber size, muscle fibers are divided into normal and atrophic fibers. In chronic processes, reinnervation to the muscle fibers is established via neighboring surviving motor neurons; therefore, an individual motor unit, consisted of a neuron and muscle fibers innervated by the neuron, undergoes enlargement. However, finally, as the number of motor neurons decreases, it results in large groups of atrophic fibers.
Muscle fibers showing neurogenic atrophy are compressed by neighboring non-atrophic fibers and show an angulated appearance on a transverse section. Atrophic fibers are usually not angulated in specimens obtained from children with spinal muscular atrophy (Fig. 8).
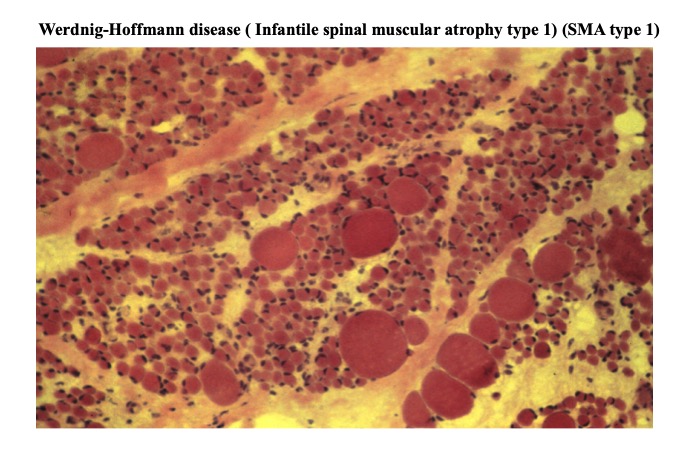
Fig.8
In neurogenic muscular atrophy, profile of atrophied fiber is not necessarily angular, as illustrated in this case of Werdnigh –Hoffmann disease (spinal muscular atrophy type I). The most important point of neurogenic change is a clear difference in diameters of atrophic and non-atrophic muscle fibers.
This could be attributed to the fact that atrophy of many fibers occurs simultaneously and rapidly without compression of neighboring fibers.
Histochemical studies are useful for the diagnosis of neurogenic changes. NADH-TR and SDH activity show a circular area of low activity at the center of the fibers, often accompanied by a band of high activity at the rim of the circular area. Fibers showing such changes are called target or targetoid fibers. Those with the high-activity band are called target fibers, and those without such a band are targetoid fibers (Fig. 9).
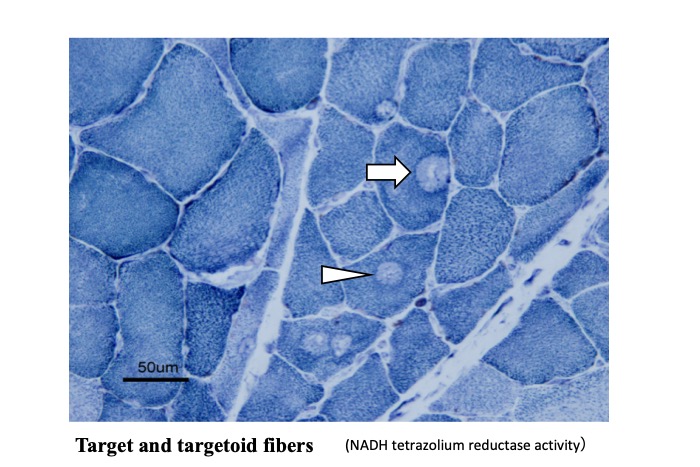
Fig.9
In denervated muscle, round areas with lower activity of NADH-tetrazolium reductase, an enzyme mainly located at mitochondria, are see in the center or epicenter of muscle fibers. Those with margin of higher activity (arrow) are target fibers, while those without clear margin (arrow head) are called targetoid fibres.
These appear in cases of acute or subacute denervation. NADH-TR and SDH activity is more intense in type 1 than in type 2 fibers. However, it should be noted that an apparent increase in enzyme activity is observed in atrophic fibers, in parallel with condensation of organelles, including mitochondria in the sarcoplasm, mimicking type 1 fiber atrophy. As neurogenic atrophy progresses, re-innervation of the denervated muscle fibers by the neighboring motor unit results in the formation of larger motor units innervating more muscle fibers than were innervated previously. All muscle fibers innervated by a motor neuron show the same fiber type; therefore, re-innervation results in fiber-type grouping (fibers of the same fiber type form a large group) (Fig. 10).
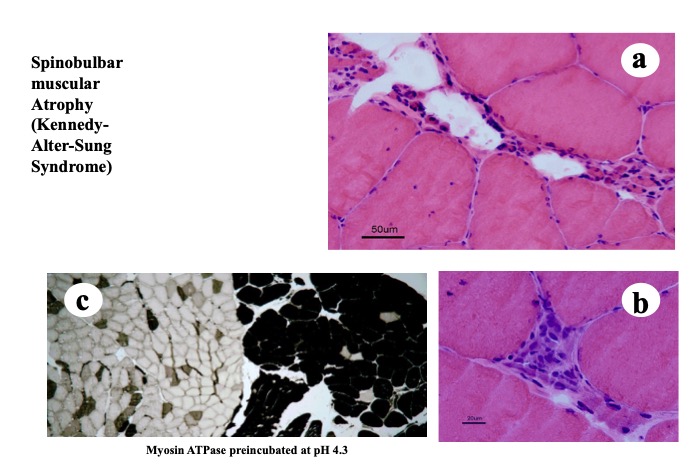
Fig.10
In chronic neurogenic atrophy of muscle, as seen in this case of bulbospinal muscular atrophy, extremely atrophic fibers and hypertrophic fibers are observed (a, b). Fiber type grouping is also seen (c).
Therefore, identification of fiber-type grouping in a muscle biopsy specimen suggests a gradually progressive neurogenic process. In some cases, chronic mild neurogenic changes are diagnosed based only on fiber-type grouping.
It is noteworthy that long-standing neurogenic atrophy often shows changes that mimick myopathic changes. These changes include increased numbers of internal nuclei, a disordered internal structure of muscle fibers, and hypertrophy of some fibers, which often occur in association with markedly atrophic fibers. Neurogenic atrophy of muscle fibers occurs in a wide variety of conditions including motor neuron diseases and peripheral neuropathies. However, a morphological distinction is often difficult.
2.Myopathic changes
Myopathies occur in a wide range of conditions; however, basic histopathological changes are common to most conditions and these are summarized as follows: (Fig. 13)
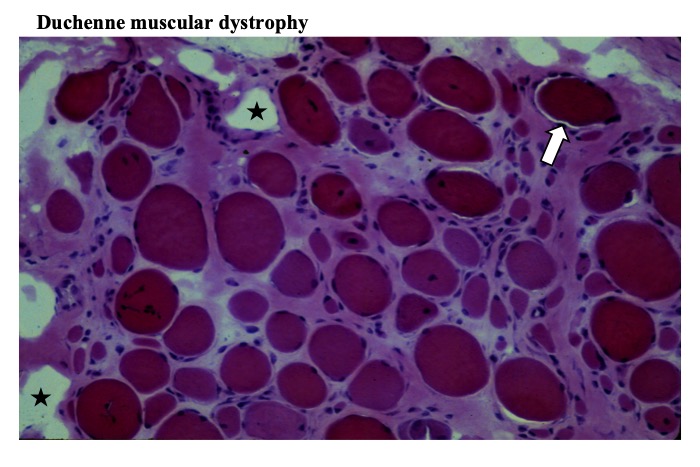
Fig.13
Muscle fibers have round contour, increased number of internal nuclei and variability in fiber diameter. Interstitial tissue is widened with fibrosis, and contain many empty spaces (stars) representing fatty deposition. Some muscle fibers called opaque fibers show dark cytoplasm (arrow) (HE stain).
(1) Evenly distributed muscle fiber diameters
Muscle fibers are evenly distributed, ranging from small to normal-diameter fibers. This even distribution differs from that observed in cases of neurogenic atrophy where fiber diameter is usually classified as small or normal.
(2) Rounded contour of muscle fibers
The contour of individual muscle fibers is affected by several factors. Simultaneous atrophy of all muscle fibers leads to a rounded contour with widened interstitial space. In contrast, when a limited number of fibers lose their volume and such fibers are interspersed among normal fibers, the atrophic fibers are compressed and appear angulated.
(3) Increased numbers of internal nuclei
Internal or central nuclei are commonly observed in myopathies; however, they may also be identified in cases of chronic neurogenic atrophy. Internal nuclei occur in <3% of normal muscle fibers, although their numbers may increase in individuals with high levels of exercise. They may be increased in congenital myopathies, such as centronuclear myopathies and central core disease. Myotonic dystrophy usually shows a significantly increased number of internal nuclei and they may occasionally occur in association with fiber splitting. Increased numbers of internal nuclei in inflammatory myopathies, such as dermatomyositis (DM) and polymyositis (PM) are attributable to regenerating muscle fibers. Myonuclei in regenerating muscle fibers are often located internally, larger in size, and paler than normal nuclei. Nuclear clumps are aggregates of myonuclei that are often identified in extremely atrophic muscle fibers. They occur in both chronic neurogenic atrophy and chronic myopathies.
(4) Widening of the interstitial tissue
The endomysium is widened during the early stages of myopathy in contrast to such widening occurring later during the disease process in neurogenic atrophy. Widening of the perimysium, followed by fibrosis and fat deposition are common changes observed in chronic myopathies, as well as neurogenic atrophy.




