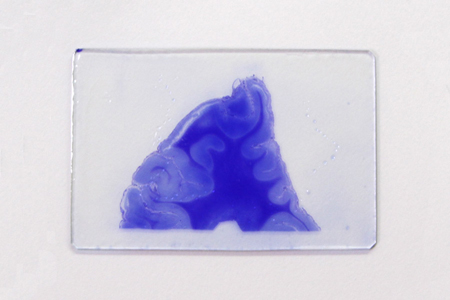
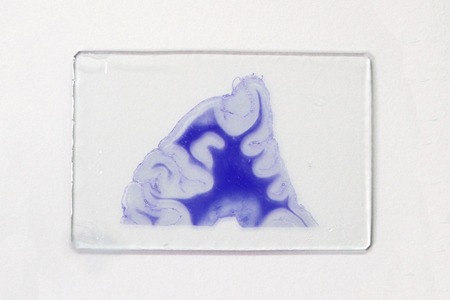
This method stains fibrous astrocytic processes blue purple with crystal violet. The proliferation of astrocyte fibrous components in response to tissue damage and gliosis can be observed topographically.
| 1 | Deparaffinization | Xylene | 3 changes, 10 minuntes each |
| 2 | Removal of xylene | 100% ethanol | 3 changes, 5 minutes each |
| 3 | Hydration | 95%, 70% ethanol | 5 minutes each |
| 4 | Mordanting *1 | Phosphomolybdic alcohol | 1 minute |
| 5 | Morderting *2 | Chloroform-alcohol mixture |
- Blot with the filter paper (four-ply) damped with chloroform-alcohol mixture, and flood with the mixture.
- Leave the section until it becomes transparent (around 10 seconds)
|
| 6 | Staining *3 | 2% crystal violet solution |
- Drain the chloroform-alcohol mixture and replace with the crystal violet solution.
- Discard the solution and blot well with filter paper (four-ply).
|
| 7 | Washing *4 | 10% pottasium bromide solution | More than 1 minute |
| 8 | Differentiation *5 | Aniline-xylene mixture |
- Blot with the filter paper (four-ply) damped with aniline-xylene mixture, and flood with the mixture.
- Change the liquid approximately every 15 minutes.
|
| 9 | Microscopic check | | Continue differentiation until only the glia fibers appear stained. |
| 10 | Washing *6 | Xylene | 2 changes, 1 minutes each |
| 11 | Clearing *6 | Xylene | 3 changes, 10 minuntes each |
| 12 | Coverslipping *6 | | |